By Miriam Raftery
October 28, 2014 (San Diego's East County) - An ABC News investigation that includes 10 News in San Diego reveals that the maker of the Essure birth control device covered up evidence that patients have been seriously harmed by the device for over a decade.
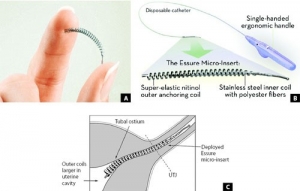
The device consists of metal coils that are implanted inside a woman’s Fallopian tubes to provide permanent birth control.
But the coils have punctured the Fallopian tubes in some women, causing miscarriages and other serious problems. In at least 91 cases, women had to undergo hysterectomies and remove their reproductive organs. In other cases, the devices have caused ectopic pregnancy, a potentially life-threatening condition in which a fertilized egg implants inside the Fallopian tube, not the uterus.
Essure has also been known to migrate out of the Fallopian tubes causing other damage including bowel perforation.
One woman said it felt like her insides were being “shredded” and that she was in constant pain. Pelvic pain, along with bloating and rashes, are common complaints among women who have had the Essure device implanted.
At least one death has occurred in a woman who suffered a strep infection that destroyed her reproductive organs after the device was implanted.
The device has also been linked to serious harm in women with allergies to nickel.
Conceptus, which made the device until the company recently sold to Bayer, was cited multiple times by the Food and Drug administration for failing to report complaints.
Thousands of complaints have occurred through the years, even triggering a Congressional hearing where women called for the device to be banned from the market. Of those, 168 complaints received by the company since 2007 were regarding perforations and other serious problems – yet Conceptus only told the FDA about 22 of those complaints.
A Facebook page now has attracted over 3,000 women describing their negative experiences with the device and Erin Brokovich, the activist who won a major lawsuit over the poisoning of a town’s water supply, has joined the fight to protect women from Essure, setting up a website for women harmed by Essure to tell their stories.
Astonishingly, women who have been harmed by the device can’t sue the manufacturer of Essure, since An FDA rule protects the manufacturer and prevents patients from suing, even though Conceptus covered up the high numbers of patients seriously harmed by its product.
Conceptus sold to Bayer HealthCare in June 2013. Bayer has not received any notices of reporting violations from the FDA. Bayer has said that it takes all adverse events seriously, but that most of the side effects to Essure are listed in the product’s instructions for use information provided to patients. But other than improving its reporting of adverse effects, has Bayer taken any steps to actually make the device safer?
ABC News sought to obtain additional details. But some records obtained from the FDA were blacked out. The TV network is now appealing those redactions.
So for now, it’s buyer beware for women who use the Essure birth control devices.







Recent comments